  In case of this change in a reaction the symbol will become ΔH⁰ᵣ. The symbol of standard enthalpy change is Delta H nought or H. These standard states are also denoted as “reference states”. It refers to a change in enthalpy that occurs in a reaction taking place under standard conditions and where the reactants are in a standard state. This change in enthalpy is represented by ΔH. Therefore, it requires some energy to break the bonds, and in return, some energy is released as well after the product is formed. It happens because, during a chemical reaction, some bonds of reactions need to be broken to produce the product. Hence, its own energy content gets low, according to the fundamental concept of energetics. The reason behind it is if a system participates in a reaction, it releases energy. Also, it is concluded that if the enthalpy decreases, a reaction is successful. The change of enthalpy in a reaction is almost equivalent to the energy gained or lost during a reaction. 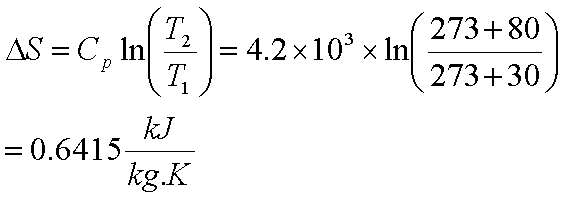
Where E is enthalpy, U is the internal energy of any system, P is pressure, and V is volume. The enthalpy is represented through the following equation. There are some molecules that take part in this change are called “internal enthalpy” and the molecules that do not are referred to as “external enthalpy”. For example, it increases when heat is added and decreases when heat is withdrawn from that system. Thereby, it changes when heat enters or leaves a system. It deals with the heat contained in any system. Relying on these two factors, a new product is formed through a standard reaction of several compounds.Įnthalpy is defined as a change in internal energy and volume at constant pressure. Both of them are partly related to each other in a reaction because the fundamental rule of any reaction is releasing or absorbing heat or energy. You just have to work out the expression for dS and integrate from T1 to T2 to get ΔS body.Enthalpy and Entropy are two significant terms related to thermodynamics. Each step would result in a heat flow dQ = mCdT. Entropy change calculator series#A reversible process for this would be to successively put it in contact in series with an arbitrarily large number of heat baths - each of which are at an infinitesimally higher temperature than the body - until it reaches T2. The solid body changes temperature from T1 to T2. 
The reversible path would simply be a reversible isothermal heat flow out of the heat bath of Q, where Q is the amount of heat flow into the solid body. Here you are assuming that the bath has an arbitrarily large heat capacity so its temperature before and after the process is the same. 
Since the actual process was NOT reversible, the reversible paths will not be the same path for each component. You just have to find a reversible path for each component (ie the solid body and the bath). the direction of heat flow) by making an infinitesimal change in conditions.īut to find the change in entropy of each body, you only look at the initial and final states - you do not care about the process that occurred in going from the initial to final states. The process is not reversible because you cannot change the direction of the process (ie. I try to solve this problem by constructing 1 isotherm and 1 adiabat to replace original isochoric path and find out that dS actually = dQ / T = Cv/T dT ,Īlthough i know where this come from ( dS = dQ /T) from maths wayīut I think i misunderstand some physical conceptĬan someone explain why dS = dQ/T, even when this process is not reversible in some physical way? There comes my question, we find out that this isochoric process is irreversibleīut when we use the equation dS = dQ/T, we could only use it in reversible process. So total entropy is not zero when T2 and T1 has a finite change. Then i compute the entropy change of heat body in isochoric system = nR ln(T2/T1)Īnd entropy change of heat bath = - Cv(T2-T1)/T2 ( i read these from notes actually but i am confused with the reversibility in it)īut I remember that we have proved beforeĭS >= dQ/T for all process, and equality hold when process is reversible(like isotherm and adiabats) We could calculate its entropy change(heat body) from dS = Cv/T dTĪnd entropy change of heat bath from dS = dQ / T2, using dQ = Cv dT in 1st equation In an isochoric process ( a solid heat body with T1 temp. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
